

Frequently asked questions about NEUPRO® (rotigotine transdermal system)
-
1. Can I get my NEUPRO Patch wet?
Yes. You can bathe, shower, or swim while wearing the NEUPRO Patch. However, water may loosen your patch. Avoid hot baths, as the heat may cause too much medicine to pass through the skin.
-
2. What if my NEUPRO Patch falls off?
If your patch falls off, put on a fresh patch until the time you usually change your patch. Then put a new patch on a different part of your skin at your regular scheduled time. If the edges of the patch lift, you may tape them down with bandaging tape.
See if you can save on NEUPRO
Eligible commercial patients may pay as little as $10 per 30-day supply of NEUPRO.*
-
3. What if I forget to put on my NEUPRO Patch?
If you miss a dose or forget to change your NEUPRO Patch, apply a new NEUPRO Patch as soon as you remember. Replace the NEUPRO Patch at your normal time the next day.
-
4. How long does it take for the NEUPRO Patch to start working?
Your doctor should start you on a low dose of NEUPRO. Your doctor may change the dose weekly until you are taking the right amount of medicine to control your symptoms. It may take several weeks before you reach the dose that controls your symptoms best.
-
5. What should I avoid while taking NEUPRO?
Do not drive, operate machinery, or do other dangerous activities until you know how NEUPRO affects you.
Avoid exposing the NEUPRO Patch you are wearing to heating pads, electric blankets, heat lamps, saunas, hot tubs, heated waterbeds, and direct sunlight. Too much medicine could be absorbed into your body.
Do not use NEUPRO during certain medical procedures called magnetic resonance imaging (MRI) or cardioversion. Using NEUPRO during these procedures could cause a burn to the site where you applied your NEUPRO Patch.
-
6. Do I have to apply my NEUPRO Patch around mealtime?
No. Apply your patch when it's convenient for you, and apply it at the same time each day. Because NEUPRO is delivered through the skin, food should not affect the medication.
-
7. Why do I need to hold the NEUPRO Patch on for at least 30 seconds?
Press your NEUPRO Patch firmly with the palm of your hand for 30 seconds to make sure there is good contact with your skin, especially around the edges. The warmth of your hand helps the adhesive on the patch stick to your skin. Make sure that your NEUPRO Patch is flat against your skin. There should be no bumps or folds in your NEUPRO Patch.
-
8. How can I prevent skin irritation or other skin-related side effects?
You need to change the location of the NEUPRO Patch on a daily basis and should not use the same location for 14 days. Changing the location will reduce the chance of skin irritation. You may see mild redness, like when you remove an adhesive bandage, which is normal. Of course, always check with your doctor if you have any concerns.
Patients should report application site reactions that do not go away after a few days, that get worse, or that spread outside the patch site. If there is a skin rash or irritation from the patch, direct sunlight on the area should be avoided until the skin heals. Exposure could lead to changes in skin color.
The NEUPRO Patch Placement Tracker can help you rotate your application site every day.
Download the Tracker now.
-
9. How do I safely stop using the NEUPRO Patch?
Do not stop or change your treatment with NEUPRO without talking to your doctor.
-
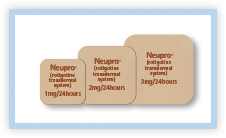
10. What does the NEUPRO Patch look like?
NEUPRO is a small, thin patch that comes in three sizes based on dosage, 1 mg, 2 mg, and 3 mg.
Is RLS affecting your life?
Some people have to live their lives around their RLS symptoms. Do you?
-
11. How can I save on NEUPRO?
The NEUPRO Patient Savings Program™* is here to help.
Check your eligibility. If you qualify, you can download your Savings Card right away.*Eligibility terms and conditions apply.
Please call ucbCARES® at 1-844-599-CARE (2273) or email ucbCARES@ucb.com if you have questions about how to apply the NEUPRO Patch.
